CAR-T cell therapy (Chimeric Antigen Receptor T-cell therapy) is one of the most significant advances in cancer treatment in recent years. This innovative form of immunotherapy uses a patient’s own immune cells to recognize and destroy cancer cells.
Initially developed for patients who had exhausted all other treatment options, CAR-T therapies have shown remarkable results in certain blood cancers. As of 2026, several CAR-T treatments have received approval from the U.S. Food and Drug Administration (FDA), and many more are currently being studied in clinical trials.
FDA-Approved CAR-T Cell Therapies (2026)
Below are the CAR-T therapies currently approved by the U.S. Food and Drug Administration (FDA).
Kymriah (tisagenlecleucel)
Approved in 2017, Kymriah was the first CAR-T therapy ever approved by the FDA.
Indications include:
- B-cell acute lymphoblastic leukemia (ALL) in pediatric and young adult patients
- Relapsed or refractory diffuse large B-cell lymphoma (DLBCL)
- Follicular lymphoma
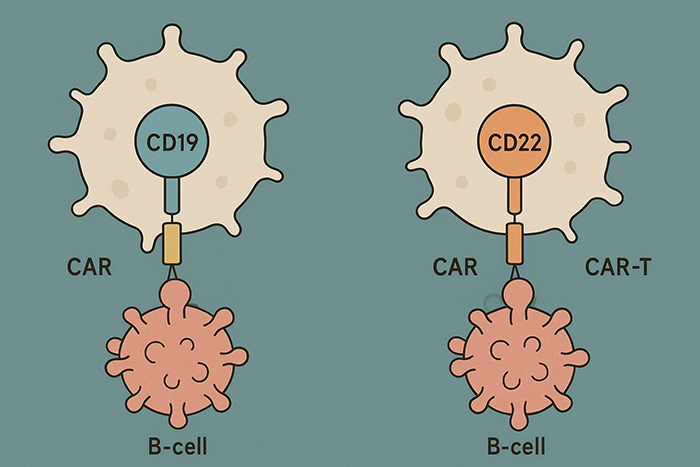
Kymriah targets the CD19 antigen, which is present on many B-cell cancers.
Yescarta (axicabtagene ciloleucel)
Approved in 2017, Yescarta is widely used for aggressive B-cell lymphomas.
Indications include:
- Diffuse large B-cell lymphoma (DLBCL)
- Primary mediastinal B-cell lymphoma
- High-grade B-cell lymphoma
- Follicular lymphoma (later lines)
Yescarta is another CD19-targeted CAR-T therapy.
Tecartus (brexucabtagene autoleucel)
Tecartus was approved in 2020 and expanded in later years to treat additional cancers.
Approved for:
- Mantle cell lymphoma
- B-cell acute lymphoblastic leukemia in adults
Like several earlier CAR-T therapies, Tecartus targets CD19.
Breyanzi (lisocabtagene maraleucel)
Breyanzi received FDA approval in 2021, with expanded approvals afterward.
Used for:
- Diffuse large B-cell lymphoma (DLBCL)
- Large B-cell lymphoma
- Chronic lymphocytic leukemia (in some settings)
Breyanzi was designed with a defined composition of CD4 and CD8 T cells, which may help improve safety and consistency.
Abecma (idecabtagene vicleucel)
Approved in 2021, Abecma was the first CAR-T therapy for multiple myeloma.
Indication:
- Relapsed or refractory multiple myeloma
Abecma targets BCMA (B-cell maturation antigen), a protein commonly expressed on myeloma cells.
Carvykti (ciltacabtagene autoleucel)
Approved in 2022, Carvykti is another BCMA-targeted therapy for multiple myeloma.
Clinical studies have shown very high response rates, including deep and durable remissions in many patients.
Carvykti is now being used earlier in the treatment sequence for eligible patients.
Aucatzyl (obecabtagene autoleucel)
Aucatzyl is one of the newer CAR-T therapies, approved for:
- Adult patients with relapsed or refractory B-cell acute lymphoblastic leukemia
This therapy is designed to improve the balance between effectiveness and safety, particularly reducing severe immune-related side effects.
Ongoing Research and Future Developments
Researchers are now working on next-generation CAR-T technologies, including:
- Dual-target CAR-T cells to prevent cancer escape
- “Off-the-shelf” CAR-T therapies created from donor cells
- CAR-T therapies for solid tumors
- Improved safety mechanisms to reduce treatment-related side effects
- Expansion of CAR-T therapy to new indications beyond blood cancers, including autoimmune and rheumatologic diseases
Early studies suggest that CAR-T therapy may help reset the immune system in certain severe autoimmune conditions such as systemic lupus erythematosus and other antibody-mediated diseases, opening the door to potential new treatment strategies.
The field continues to advance rapidly, and new CAR-T therapies and indications may receive regulatory approval in the coming years.
Sources
The information in this article is based on publicly available scientific and regulatory sources:
- U.S. Food and Drug Administration (FDA) – Cellular & Gene Therapy Products
- National Cancer Institute (NCI) – CAR T-Cell Therapy Fact Sheet
- American Society of Clinical Oncology (ASCO) – CAR-T therapy resources
- New England Journal of Medicine (NEJM) – clinical trial publications on CAR-T therapy.
- Lancet Oncology and Blood Journal – peer-reviewed studies on CAR-T treatments.
- Manufacturer information and pivotal trials for Kymriah (Novartis), Yescarta & Tecartus (Gilead/Kite), Breyanzi (Bristol Myers Squibb), Abecma (Bristol Myers Squibb), Carvykti (Johnson & Johnson / Legend Biotech)
Publication date: March 2026